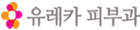JAAD
DERMATOLOGIC SURGERY
아래 내용은 JAAD에 게재된 논문내용입니다.
for refractory vitiligo: A etrospective study of 230 cases
Jung Min Bae, MD, PhD,a Ji Hae Lee, MD, PhD,a Hyuck Sun Kwon, MD,a Jiehoon Kim, MD,b
and Dong Seok Kim, MD, PhDc Suwon and Seoul, Korea
Objective: To assess the therapeutic effectiveness and adverse events associated with micropunch grafting in refractory vitiligo.
Methods: We retrospectively reviewed 230 cases in 208 patients with stable vitiligo who underwent motorized 0.8-mm micropunch grafting during January 2015-August 2017. Treatment success was defined as ≥75% repigmentation, and factors associated with the outcome were assessed.
Results: Overall, 181 of 230 lesions (78.7%) achieved treatment success after a median of 6 months with postoperative excimer therapy. Lesions on the face and neck, and disease stability of ≥12 months were good prognostic factors for treatment success. Common adverse events were color mismatch (24.8%) and cobblestone appearance (18.3%). Overall, the treatment was tolerable.
Limitations: This was a retrospective study.
Conclusion: Micropunch grafting using a motorized 0.8-mm punch can successfully treat refractory vitiligo with short procedure times and excellent outcomes. This technique could be a rapid and convenient surgical option with acceptable adverse events and is promising for treating refractory vitiligo on an outpatient basis, particularly in patients who are unlikely to tolerate prolonged surgery.
( J Am Acad Dermatol 2018;79:720-7. )
Key words: grafting, operation, punch grafting, surgery, transplantation, vitiligo.
|
Abbreviations used: CI : confidence interval OR: odds ratio UV: ultraviolet |
| From the Department of Dermatology, St. Vincent’s Hospital, College of Medicine, The Catholic University of Korea, Suwona; Dr Kim’s Skin & Laser Clinic, Suwonb; Eureka Skin & Laser Clinic, Seoul.c Drs Bae and Lee contributed to this work equally. Funding sources: Supported by the Catholic Medical Center Research Foundation in program year 2017. Conflicts of interest: None disclosed. |
Correspondence to: Jung Min Bae, MD, PhD, Department of Dermatology, St. Vincent’s Hospital, 93, Jungbu-daero, Paldal-gu, Suwon, 16247, Korea. E-mail: jminbae@gmail.com; Dong Seok Kim, MD, PhD, Eureka Skin & Laser Clinic, 101 Teheran-ro, Gangnam-gu, Seoul, 06134, Korea. E-mail: dongkim3@hotmail.com. 0190-9622/$36.00 ⓒ2018 by the American Academy of Dermatology, Inc. https://doi.org/10.1016/j.jaad.2018.06.016 |
| Accepted for publication June 12, 2018. Video available at http://www.jaad.org. Reprints not available from the authors. |
|
Vitiligo is a long-term skin condition characterized by white patches with selective loss of melanocytes.1 It affects 1% of the population worldwide and can have a profound impact on quality of life.2,3 Although phototherapy, including narrow-band ultraviolet (UV) B and excimer laser treatments, has been widely used to treat vitiligo, nonsurgical treatments are not always successful.4 Thus, surgical treatment would be a viable option for patients with refractory vitiligo.5 Surgical techniques for vitiligo include punch grafting, suction blister grafting, and cellular grafting.6 Punch grafting, a method of directly transferring 1e2-mm punch grafts from donor to recipient sites,7 is easy to perform and does not require any specialized equipment. However, its adoption has been limited as it is time-consuming and can lead in some cases to a cobblestone appearance. Suction blister grafting, ie, taking intact epidermis from the donor site using negative pressure and transplanting it to the recipient site, has been used for the treatment of vitiligo, but this method is also time-consuming, difficult to use when treating large lesions, and associated with adverse events, such as color mismatch and perilesional halo.8 Cellular grafting using cultured or noncultured suspensions of epidermal cells has the advantage of being able to treat large recipient sites with small amounts of donor tissue.9 However, this technique requires a long procedure time and special laboratory equip- ment in addition to a skilled team. While there are pros and cons for each of these surgical techniques, punch grafting has a number of advantages, particularly for the treatment of small lesions. We developed a micropunch grafting technique involving the application of a motorized 0.8-mm punch to overcome the disadvantages of conventional punch grafting. We performed a retrospective review of vitiligo patients treated with this technique to evaluate its effectiveness and examine factors associated with treatment success and adverse events.
METHODS
Study design and setting This was a retrospective interventional case-series study. Medical records and photographs of 230 cases in 208 patients with refractory vitiligo who underwent motorized 0.8-mm micropunch grafting during January 2015-August 2017 in our clinics were retrieved and analyzed. Grafting was performed for stable vitiligo lesions that had not improved with nonsurgical treatment for 3 months. The stable vitiligo lesion was defined as no progression for >3months. We included patients who received excimer laser treatment for at least 3 months after grafting to assess the treatment response. This study was approved by the institutional review board of St. Vincent’s Hospital (VC17RESI0108).0.8-mm motorized micropunch grafting
A stainless-steel punch 0.8 mm in diameter loaded into the handpiece of a micromotor (SST-C1, Ilooda Co, Suwon, Korea) was used for skin grafting from both donor and recipient sites. The micromotor was adjusted to a speed of 900-1200 rotations/min, with a torque-to-speed ratio of 1:8. The donor sites were the back of the ear or the postauricular region. Both recipient and donor sites were cleaned with antiseptic solution followed by injection of 2% lidocaine with epinephrine at a concentration of 1:200,000. At recipient sites, the skin was removed to create chambers for planting grafts at intervals of 4 mm using the motorized micropunch. At donor sites, the grafts were harvested at intervals of 1 mm using the motorized micropunch. The grafts were handled with curved jeweler forceps and then placed in recipient chambers. Hemostasis was achieved on both sites by applying gentle pressure and covering with sterile gauze. Hydrocolloid and Steri-Strip (3M, Maplewood, MN) dressings were applied to the donor and recipient sites, respectively, without suturing for 1 week.Postsurgery excimer laser treatment
One week after surgery, all patients were treated with a 308-nm xenon chloride excimer laser (E1, Jetema, Seoul, Korea) on 2 nonconsecutive days per week. The dose was initiated at 100 mJ/cm2 and increased by 50 mJ/cm2 at subsequent sessions until pink erythema appeared and persisted for 24 h. Topical 0.1% tacrolimus ointment (Protopic, Leo Pharma Inc, Ballerup, Denmark) was applied twice weekly to all surgical sites throughout the laser treatment period.Covariates
Age, sex, body site, disease duration, size, disease stability, postoperative time, and subtype (focal, segmental, and nonsegmental) were used asexplanatory variables. Disease stability was defined as a period with no new lesions or no progression of previous lesions.10 The postoperative time was defined as the time of the ast follow-up in the medical records.Treatment responses
Treatment responses were evaluated on the basis of the clinical photographs obtained at baseline and on follow-up visits. Two dermatologists (Drs Lee and Kwon) compared the initial pretreatment photo- graphs with the final follow-up photographs. Treatment success was defined as ≥75% repigmentation of the surgery site.5 Repigmentation of 50%- 74% was defined as partial response and <50% as insufficient repigmentation.Adverse events
The occurrence of any adverse event was deter- mined from the medical records and confirmed with clinical photographs. We further investigated cobblestone appearance, color mismatch, hyperpig- mentation, and perilesional halo and evaluated each in 4 grades according to severity (grade 0 none, grade 1 somewhat, grade 2 obvious, and grade 3 pronounced). Of these, grades 2 and 3 were consid- ered adverse events. Other adverse events, such as infection, scarring, keloid and milia formation, or tire-track appearance from both donor and recipient sites were also monitored.Time required for surgery
The time taken for surgery was measured pro- spectively in a total of 42 patients. Surgery time was defined as the time taken from the start of surgery after anesthesia to completion of grafting.Statistical analyses
Univariable and multivariable logistic regression models were used in sequence to determine the independent factors associated with outcomes, including treatment success, insufficient repigmen- tation, and each adverse event. In all analyses, P < .05 was taken to indicate statistical significance. All analyses were performed using R 3.3.1 (R Foundation for Statistical Computing, Vienna, Austria).RESULTS
A total of 230 lesions in 208 patients (female 47.6%, median age 32.7 years, range 5-68 years) were included in the study (Table I). The median duration of vitiligo before grafting was 9.5 years (range 6 months-47 years). Segmental vitiligo (59.1%) was the most common, followed by nonsegmental vitiligo (30.3%), and focal vitiligo (10.6%). The face and neck (70.4%) was the most common site; other sites included the extremities (11.3%), trunk (10.9%), and hands and feet (7.4%). According to the size of the recipient site, there were 139 (60.4%) lesions ≤9cm2 in size, 64 (27.8%) lesions 10-49 cm2 in size, 7(3.0%) lesions 50-99 cm2 in size, and 20 (8.7%) lesions ≥100 cm2 in size.Table I. Clinical characteristics of vitiligo patients enrolled in this study
|
Characteristics |
value,n(%) |
|---|---|
|
Total patient |
205(100) |
|
sex |
|
|
Male |
109(52.4) |
|
Female |
99(47.6) |
|
Age, y |
|
|
<20 |
36(17.3) |
|
20-39 |
115(55.3) |
|
40-59 |
47(22.6) |
|
≥60 |
10(4.8) |
|
Subtype |
|
|
Focal vitiligo |
22(10.6) |
|
Segmental vitiligo |
123(59.1) |
|
Nonsegmental vitiligo |
63(30.3) |
|
Disease duration, y |
|
|
≤1 |
10(4.8) |
|
1-4 |
57(27.4) |
|
5-9 |
64(30.8) |
|
≥10 |
77(37.0) |
| Total operation cases | 230(100) |
|
Body part |
|
|
Face and neck |
162 (70.4) |
|
Trunk |
25 (10.9) |
|
Extremities |
26 (11.3) |
|
Hands and feet |
17 (7.4) |
|
Size, cm |
|
|
<9 |
139 (60.4) |
|
10-49 |
64 (27.8) |
|
50-99 |
7 (3.0) |
|
≥100 |
20 (8.7) |
|
Disease stability, mo |
|
|
<6 |
25 (10.9) |
|
6-11 |
39 (17.0) |
|
12-23 |
49 (21.3) |
|
≥24 |
117 (50.9) |
|
Postoperative time, mo |
|
|
<6 |
124 (53.9) |
|
6-11 |
62 (27.0) |
|
≥12 |
44 (19.1) |
|
CAPSULE SUMMARY Punch grafting for vitiligo is time consuming and could result in a cobblestone appearance. Micropunch grafting using a motorized 0.8-mm punch yields excellent results with a short operation time. Micropunch grafting represents a convenient surgical option for refractory vitiligo. |

| Fig 1. Repigmentation after micropunch grafting in refractory vitiligo to nonsurgical treatment. A-D, A 10-year-old girl with segmental vitiligo on the forehead before surgery (A), just after surgery (B), 2 months after surgery with subsequent excimer laser treatments (C), and 6 months after surgery (D). The cobblestone appearance, which was observed 2 months after surgery, was improved after 6 months. E and F, A 54-year-old woman with nonsegmental vitiligo on the neck before surgery (E) and 6 months after surgery (F). G and H, A 47-year-old man with nonsegmantal vitiligo on the hand before surgery (G) and 6 months after surgery (H). |
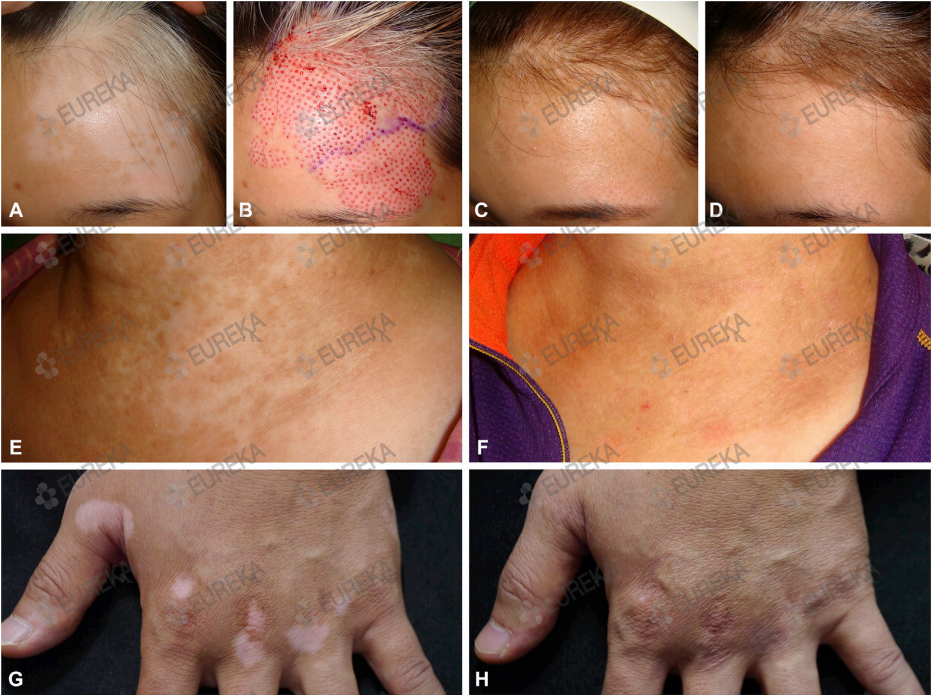
Treatment success The overall rate of treatment success (≥75% repigmentation) was 78.7% (Fig 1); 67.4% of the patients showed almost complete (≥90%) repigmen- tation. Treatment success was observed in 89.5% on the face and neck, 65.4% on the extremities, 52.0% on the trunk, and 35.3% on the hands and feet (Fig 2). In multivariable analyses, the face and neck and disease stability ≥12 months were identified as independent factors associated with good response to micropunch grafting (Table II).
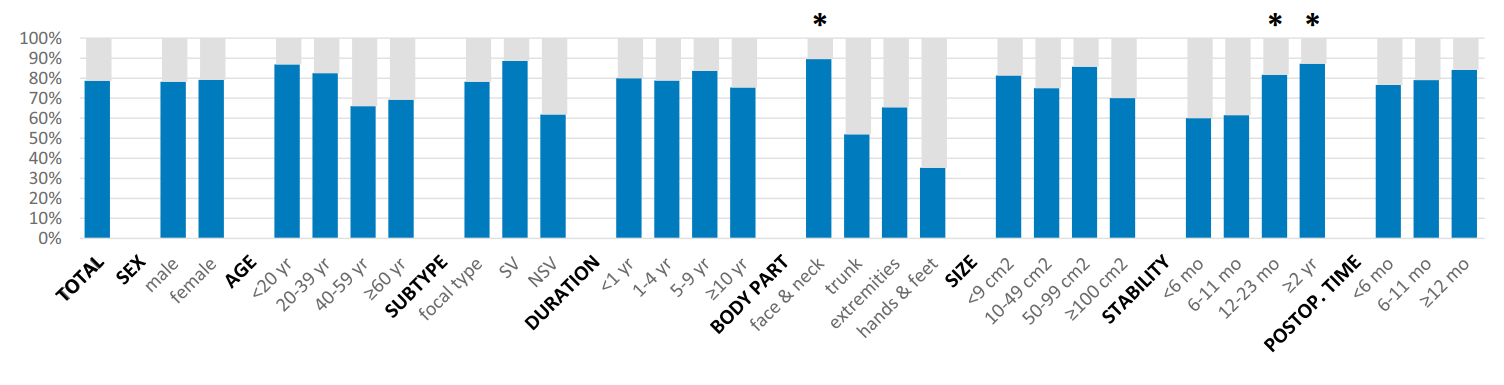
Insufficient repigmentation Insufficient repigmentation was observed in 12.6% of cases at rates of 6.2% for face and neck lesions, 28.0% for the trunk, 19.2% for the extrem- ities, and 41.2% for the hands and feet. Nonsegmental vitiligo subtype (odds ratio [OR] 6.079, 95% confidence interval [CI] 1.958-21.687, compared with segmental vitiligo), hands and feet (OR 4.072, 95% CI 1.025-16.557, compared with face and neck), and disease stability <12 months (<6 months, OR 7.713; 6-11 months, OR 3.003; compared with ≥24 months) were highly correlated with insufficient repigmentation (Table II).
Adverse events Color mismatch was the most common adverse event after micropunch grafting for 57 lesions (24.8%). A cobblestone appearance was observed in 42 (18.3%) cases, hyperpigmentation in 26 (11.3%), and perilesional halo in 14 (6.1%) (Fig 3, Supplemental Table I; available at http://www.jaad. org). Color mismatch was significantly prevalent on hands and feet (OR 9.432, compared with face and neck) and decreased gradually with time after surgery (P for trend <.001). The prevalence of a cobblestone appearance was significantly higher in younger patients (<20 years of age) and decreased in elderly patients (P for trend .012) and improved gradually with time after surgery (P for trend .045) (Fig 4, Supplemental Table I). None of the patients had postsurgical infections, and no scarring devel- oped on either donor or recipient sites.
INSUFFICIENT REPIGMENTATION (<50% repigmentaon) |
TREATMENT SUCCESS (≥75% repigmentaon) |
| Fig 2. Treatment responses, by various factors. *Factor significantly associated with outcome (P < .05). NSV, Nonsegmental vitiligo; postop, postoperative; SV, segmental vitiligo. |
Time required for surgery The median surgery time measured in 42 cases was 28 minutes. It varied depending on the size of the recipient lesion, with medians of 17 minutes for lesions <9cm2, 42minutes for those of 10-49 cm2, and 72minutes for those of 50-99cm2.
DISCUSSION Surgical treatment of leukoderma using autologous punch grafts was first reported by Orentreich in 1972,11 and then Falabella discovered that a 1-mm donor graft could produce an area of pigmentation that was 25 times larger than its size, providing the basis for punch grafting in vitiligo.12,13 Punch grafting is a commonly used surgical technique for refractory vitiligo, and punch grafts of 1.5 mm, 2 mm, or 2.5 mm have conventionally been used for the surgery.14 When graft diameters are <1.2 mm, some authors use the term minigrafting interchangeably with punch grafting.13,15 Previous studies support the high effectiveness of punch grafting, with success rates of 67%-87%.7,16-18 Punch grafting has been combined with psoralen plus UV-A or narrow-band UV-B phototherapy, resulting in better therapeutic outcomes.18,19
Although punch grafting is the simplest and least expensive surgical technique for treating refractory vitiligo, it has limitations. It can be time-consuming for large vitiligo areas, so only small lesions are usually treated. In addition, a cobblestone appearance is a potential adverse event, occurring in 21%- 33% of cases on both donor and recipient sites.20 Thus, suction blister grafting and cellular grafting, which can effectively treat larger areas, are increas- ingly being adopted. However, both of these pro- cedures are often difficult to perform because the long procedure time required for the formation of blisters or cell processing. Punch grafting might still be the best procedure in outpatient clinics to treat small remaining lesions that have not fully recovered after phototherapy. Furthermore, it can be applied effectively to convex or uneven areas and areas with many movements, such as the eyelids or perioral area, where cellular grafting is difficult.
We have shortened the long procedure time using a motorized 0.8-mm micropunch that was originally used for follicular unit extraction hair transplantation. In the present study, we used the motorized micropunch device to harvest the graft from the donor site and create chambers at the recipient site. Our technique showed a notable treatment success rate of 78.7% for refractory lesions that did not respond to conventional phototherapy, and the adverse events were minimal and mostly tolerable.
Motorized 0.8-mm micropunch grafting for viti- ligo has several advantages. First, it markedly re- duces the time required for surgery, as it is possible to harvest many grafts within a short period of time. We prospectively measured the surgery times in 42 patients and found that it was not too long for young patients to withstand surgery. Our procedure is much faster than suction blister grafting and cellular grafting, which takes 2-3 hours to obtain blister grafts or to process cell suspension preparations. Second, use of a small punch 0.8 mm in diameter reduces the risk for a cobblestone appearance. For our cases, the overall incidence of cobblestone appearance was 18.3%, which decreased to 11.4% after 1 year, and was an acceptable adverse event in most cases. No scarring was observed at either donor or recipient sites. Third, the motorized punch makes it possible to form uniform, fine grafts and recipient chambers, while manual punch grafting could make grafts and chambers of uneven shapes and depth, leading to the likelihood of a cobblestone appearance.15 Fourth, simple postoperative dressings, such as Steri-Strip, allowed patients to return to normal life immediately after surgery. In addition, this proced- ure is particularly promising for patients who cannot tolerate long surgeries, as this technique can be performed within about 30 minutes.
In our study, lesions on the face and neck and disease stability of ≥12 months were good prog- nostic factors for treatment success. On the other hand, the overall rate of insufficient repigmentation was 12.6%, and was highly associated with vitiligo of the hands and feet, nonsegmental vitiligo subtype, and disease stability <12 months. Kato et al reported that patients <15 years of age with facial grafts and the segmental subtype showed better results with punch grafting.21 Feetham et al reported that patients <20 years of age had better repigmentation than older patients and that the neck and trunk showed the greatest rates of repigmentation.18 In the present study, we also found higher treatment success rates in young patients <20 years of age compared with the older age groups, but this finding was not statistically significant.
Color mismatch and a cobblestone appearance were the most common adverse events after micro- punch grafting. The rate of color mismatch was higher on the hands and feet, and the rate of having a cobblestone appearance was higher for patients <20 years of age. All adverse events improved with time after surgery, and therefore, the long-term results would probably be better than our data.
This study had some limitations. First, it was a retrospective study, in which patients had different postoperative treatment periods. Therefore, pro- spective studies are needed to confirm our findings. Second, it is hard to evaluate the efficacy of the surgery alone because subsequent excimer laser treatment and topical tacrolimus were applied after the surgery. Third, our findings might not be generalizable to patients of different races and skin types. More studies are needed.
In conclusion, we demonstrated the effectiveness of motorized 0.8-mm micropunch grafting for re- fractory vitiligo to nonsurgical treatment. This tech- nique is rapid and convenient with acceptable rates of adverse events, even though it is still difficult to cover large areas. Given the benefits of short procedure time and excellent outcome, this method is promising for treating refractory vitiligo on an outpatient basis, particularly in patients with small lesions and in patients who are unlikely to tolerate prolonged surgery. Our data helps in the identification of vitiligo patients who would likely benefit the most from micropunch grafting and helps introduce realistic surgery outcomes.
Table II. Treatment response and associated factors of micropunch grafting (N = 230)
| Variable | % | N | Univariable analyses | Multivariable analyses | ||
|---|---|---|---|---|---|---|
| Unadjusted OR(95% CI) | P value | Adjusted OR (95% CI) | P value | |||
| Treatment success | 78.7 | 181/230 | ||||
|
Body part |
||||||
|
Face and neck |
89.5 | 145/162 | Reference | Reference | ||
|
Trunk |
52.0 | 13/25 | 0.127 (0.049-0.323 | <.001 | 0.105 (0.037-0.289) | <.001 |
|
Extremities |
65.4 | 17/26 | 0.221 (0.086-0.588) | .002 | 0.224 (0.081-0.636) | .004 |
|
Hands and feet |
35.3 | 6/17 | 0.064 (0.020-0.189) | <.001 | 0.050 (0.014-0.161) | <.001 |
|
Disease stability, mo |
||||||
|
<6 |
60.0 | 15/25 | 0.221 (0.084-0.587) | .002 | 0.159 (0.050-0.495) | .002 |
|
6-11 |
61.5 | 24/39 | 0.235 (0.100-0.546) | <.001 | 0.190 (0.070-0.499) | <.001 |
|
12-23 |
81.6 | 40/49 | 0.654 (0.268-1.667) | .356 | 0.574 (0.207-1.638) | .289 |
|
≥24 |
87.2 | 102/117 | Reference | Reference | ||
| Insufficient repigmentation | 12.6 | 29/230 | ||||
|
Subtype |
||||||
|
Focal vitiligo |
8.7 | 2/23 | 2.400 (0.329-11.961) | .314 | 1.869 (0.235-10.338) | .498 |
|
Segmental vitiligo |
3.8 | 5/131 | Reference | Reference | ||
|
Nonsegmental vitiligo |
28.9 | 22/76 | 10.267 (3.972-31.937) | <.001 | 6.079 (1.958-21.687) | .003 |
|
Body part |
||||||
|
Face and neck |
6.2 | 10/162 | Reference | Reference | ||
|
Trunk |
28.0 | 7/25 | 5.911 (1.940-17.421) | .001 | 4.092 (1.126-14.476) | .029 |
|
Extremities |
19.2 | 5/26 | 3.619 (1.045-11.281) | .031 | 2.344 (0.609-8.299) | .194 |
|
Hands and feet |
41.2 | 7/17 | 10.640 (3.283-34.340) | <.001 | 4.072 (1.025-16.557) | .046 |
|
Disease stability, mo |
||||||
|
<6 |
32.0 | 8/25 | 6.412 (2.106-19.773) | <.001 | 7.713 (2.075-30.698) | .003 |
|
6-11 |
20.5 | 8/39 | 3.516 (1.203-10.313) | .020 | 3.003 (0.912-10.088) | .069 |
|
12-2 |
10.2 | 5/49 | 1.548 (0.447-4.903) | .464 | 1.112 (0.288-4.004) | .872 |
|
≥24 |
6.8 | 8/117 | Reference | Reference | ||
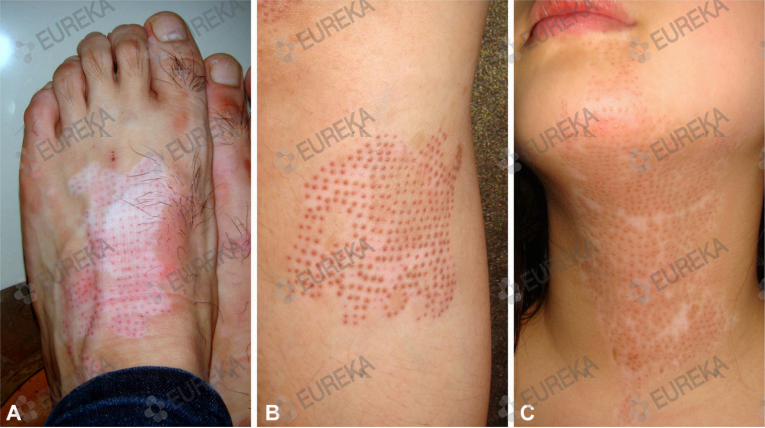
| Fig 3. Insufficient repigmentation and common adverse events of micropunch grafting. A, Insufficient repigmentation after 4 months in a 48-year-old man. B, Color mismatch between graft and surrounding skin after 6 months in a 24-year-old man. C, Cobblestone appearance after 6 months in an 18-year-old girl. |
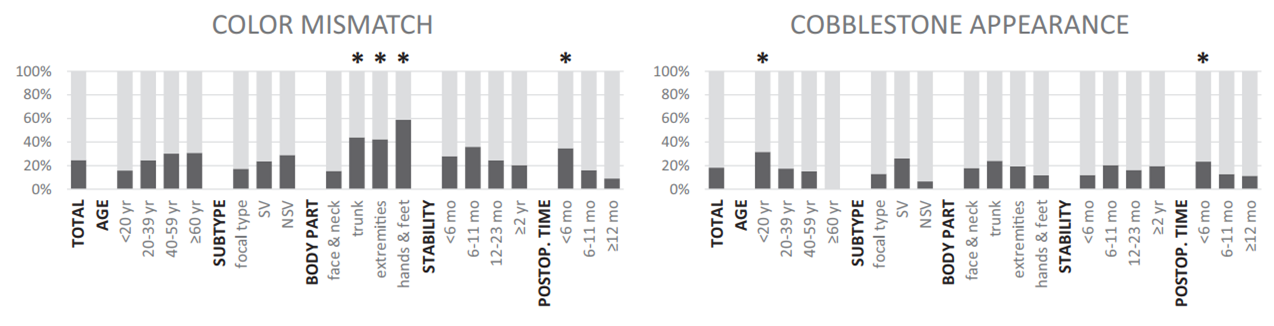
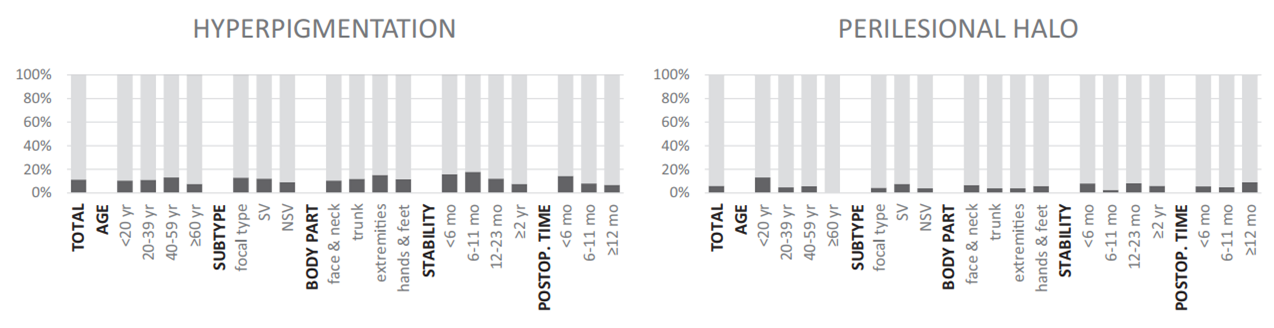
| Fig 4. Common adverse events and associated factors of micropunch grafting. *Factor significantly associated with outcome (P<.05). NSV, Nonsegmental vitiligo; postop, post-operative; SV, segmental vitiligo. |
REFERENCES
- 1. Rodrigues M, Ezzedine K, Hamzavi I, Pandya AG, Harris JE. New discoveries in the pathogenesis and classification of vitiligo. J Am Acad Dermatol. 2017;77:1-13.
- 2. Bae JM, Lee SC, Kim TH, et al. Factors affecting quality of life in patients with vitiligo: a nationwide study. Br J Dermatol. 2018; 178:238-244.
- 3. Linthorst Homan MW, Spuls PI, de Korte J, Bos JD, Sprangers MA, van der Veen JP. The burden of vitiligo: patient characteristics associated with quality of life. J Am Acad Dermatol. 2009;61:411-420.
- 4. Bae JM, Jung HM, Hong BY, et al. Phototherapy for vitiligo: a systematic review and meta-analysis. JAMA Dermatol. 2017; 153:666-674.
- 5. Rodrigues M, Ezzedine K, Hamzavi I, Pandya AG, Harris JE. Current and emerging treatments for vitiligo. J Am Acad Dermatol. 2017;77:17-29.
- 6. Mohammad TF, Hamzavi IH. Surgical therapies for vitiligo. Dermatol Clin. 2017;35:193-203.
- 7. Malakar S, Dhar S. Treatment of stable and recalcitrant vitiligo by autologous miniature punch grafting: a prospective study of 1,000 patients. Dermatology. 1999;198:133-139.
- 8. Hann SK, Im S, Bong HW, Park YK. Treatment of stable vitiligo with autologous epidermal grafting and PUVA. J Am Acad Dermatol. 1995;32:943-948.
- 9. Huggins RH, Henderson MD, Mulekar SV, et al. Melanocyte-- keratinocyte transplantation procedure in the treatment of vitiligo: the experience of an academic medical center in the United States. J Am Acad Dermatol. 2012;66:785-793.
- 10. Ezzedine K, Lim HW, Suzuki T, et al. Revised classification/no- menclature of vitiligo and related issues: the Vitiligo Global Issues Consensus Conference. Pigment Cell Melanoma Res. 2012;25:E1-E13.
- 11. Orentreich N, Selmanowitz VJ. Autograft repigmentation of leukoderma. Arch Dermatol. 1972;105:734-736.
- 12. Falabella R. Repigmentation of segmental vitiligo by autologous minigrafting. J Am Acad Dermatol. 1983;9: 514-521.
- 13. Falabella R. Repigmentation of stable leukoderma by autologous minigrafting. JDermatolSurgOncol. 1986;12: 172-179.
- 14. Mutalik S, Ginzburg A. Surgical management of stable vitiligo: a review with personal experience. Dermatol Surg. 2000;26: 248-254.
- 15. Boersma BR, Westerhof W, Bos JD. Repigmentation in vitiligo vulgaris by autologous minigrafting: results in nineteen patients. J Am Acad Dermatol. 1995;33:990-995.
- 16. Gupta S, Jain VK, Saraswat PK. Suction blister epidermal grafting versus punch skin grafting in recalcitrant and stable vitiligo. Dermatol Surg. 1999;25:955-958.
- 17. Chandrashekar B, Madura C, Varsha D. Autologous mini punch grafting: an experience of using motorized power punch in 10 patients. J Cutan Aesthet Surg. 2014;7:42-45.
- 18. Feetham HJ, Chan JL, Pandya AG. Characterization of clinical response in patients with vitiligo undergoing autologous epidermal punch grafting. Dermatol Surg. 2012;38:14-19.
- 19. Lommerts JE, Uitentuis SE, Bekkenk MW, de Rie MA, Wolkerstorfer A. The role of phototherapy in the surgical treatment of vitiligo: a systematic review. J Eur Acad Dermatol Venereol. 2018 [Epub ahead of print].
- 20. Rusfianti M, Wirohadidjodjo YW. Dermatosurgical techniques for repigmentation of vitiligo. Int J Dermatol. 2006;45: 411-417.
- 21. Kato H, Furuhashi T, Ito E, et al. Efficacy of 1-mm minigrafts in treating vitiligo depends on patient age, disease site and vitiligo subtype. J Dermatol. 2011;38:1140-1145.
Supplemental Table I. Adverse events of micropunch grafting, by various factors (N = 230)
| % | N | Univariable analyses | Multivariable analyses | |||
|---|---|---|---|---|---|---|
| Unadjusted OR(95% CI) | P value | Adjusted OR (95% CI) | P value | |||
| Color mismatch | 24.8 | 57/230 | ||||
|
Body part |
||||||
|
Face and neck |
15.4 | 25/162 | Reference | Reference | ||
|
Trunk |
44.0 | 11/25 | 4.306 (1.732-10.588) | .001 | 4.289 (1.644-11.237) | .003 |
|
Extremities |
42.3 | 11/26 | 4.019 (1.630-9.757) | .002 | 6.232 (2.340-16.987) | <.001 |
|
Hands and feet |
58.8 | 10/17 | 7.829 (2.757-23.462 | <.001 | 9.432 (3.065-31.574) | <.001 |
|
Postoperative time, mo |
<.001* | <.001* | ||||
|
<6 |
34.7 | 43/124 | Reference | Reference | ||
|
6-11 |
16.1 | 10/62 | 0.362 (0.160-0.759) | 0.258 (0.099-0.606) | ||
|
≥12 |
9.1 | 4/44 | 0.188 (0.054-0.506 | 0.140 (0.037-0.409) | ||
| Cobblestone appearance | 18.3 | 42/230 | ||||
|
Age, y |
.012* | .012* | ||||
|
<20 |
31.6 | 12/38 | Reference | Reference | ||
|
20-39 |
17.5 | 22/126 | 0.458 (0.246-1.065) | 0.463 (0.202-1.088) | ||
|
40-59 |
15.1 | 8/53 | 0.385 (0.135-1.051) | 0.400 (0.138-1.108 | ||
|
≥60 |
0 | 0/13 | 0 (NA) | 0 (NA) | ||
|
Postoperative time, mo |
.043* | .045* | ||||
|
<6 |
23.4 | 29/124 | Reference | Reference | ||
|
6-11 |
12.9 | 8/62 | 0.485 (0.195-1.093) | 0.477 (0.190-1.091) | ||
|
≥12 |
11.4 | 5/44 | 0.420 (0.135-1.082) | 0.415 (0.132-1.088) | ||